Membrane Transport Proteins
Communication between neurons is established by chemical messengers called neurotransmitters. Upon arrival of a signal (in the form of an electrical current flow) to the axon terminal of the presynaptic neuron, chemical transmission of the signal to the postsynaptic neuron takes place via a calcium-dependent release of neurotransmitters into the synaptic gap. These signaling molecules bind and activate ligand-gated ion channels and g-protein-coupled receptors on the postsynaptic neuron dendrites. Excitatory or inhibitory responses in the postsynaptic neurons are generated by the activation of these proteins. Neurotransmission is regulated by the neurotransmitter:sodium symporter (NSS) family of transporters via reuptake of excess neurotransmitters from the synapse back into the presynaptic cells or via uptake into the glial cells. The transport of neurotransmitters is performed against their concentration gradient. Hence, energy input is required for the NSS transporters to perform their function. The energy required for the transport is harnessed from the flow (co-transport) of Na+ ions down their electrochemical gradient across the membrane, being the reason why those transporters are named/called secondary transporters or symporters. Control of synaptic levels of neurotransmitters is essential to avoiding many neurological disorders and cytotoxicity. Dysfunction of NSS members has been linked to numerous neurological disorders including epilepsy, depression, anxiety, attention-deficit hyperactivity disorder (ADHD), orthostatic intolerance, and Parkinson’s disease. Hence, making them important targets for a wide range of therapeutic drugs against various neurological disorders (and also for many illicit drugs). Imipramine (Tofranil), which inhibits noradrenaline (norepinephrine) uptake, or fluoxetine (Prozac), which inhibits serotonin uptake, is an example of such clinical drugs. NSS members are also targeted by illicit drugs such as cocaine and amphetamine. Our lab explores the detailed functional mechanisms of NSS proteins, each of which is a biomolecular machine, by combining Molecular Dynamics Simulations with the principles of physics and engineering. This, in turn, provides crucial insight on how the activities/functions of these transporters can be regulated/inhibited; Granting this type of modeling an important place in drug design and development.

Related Articles:
1.
Gur, M., Golcuk, M., Yilmaz, S. Z., Taka, E. (2019).
Thermodynamic first law efficiency of membrane proteins.
Journal of Biomolecular Structure & Dynamics, 1-11.
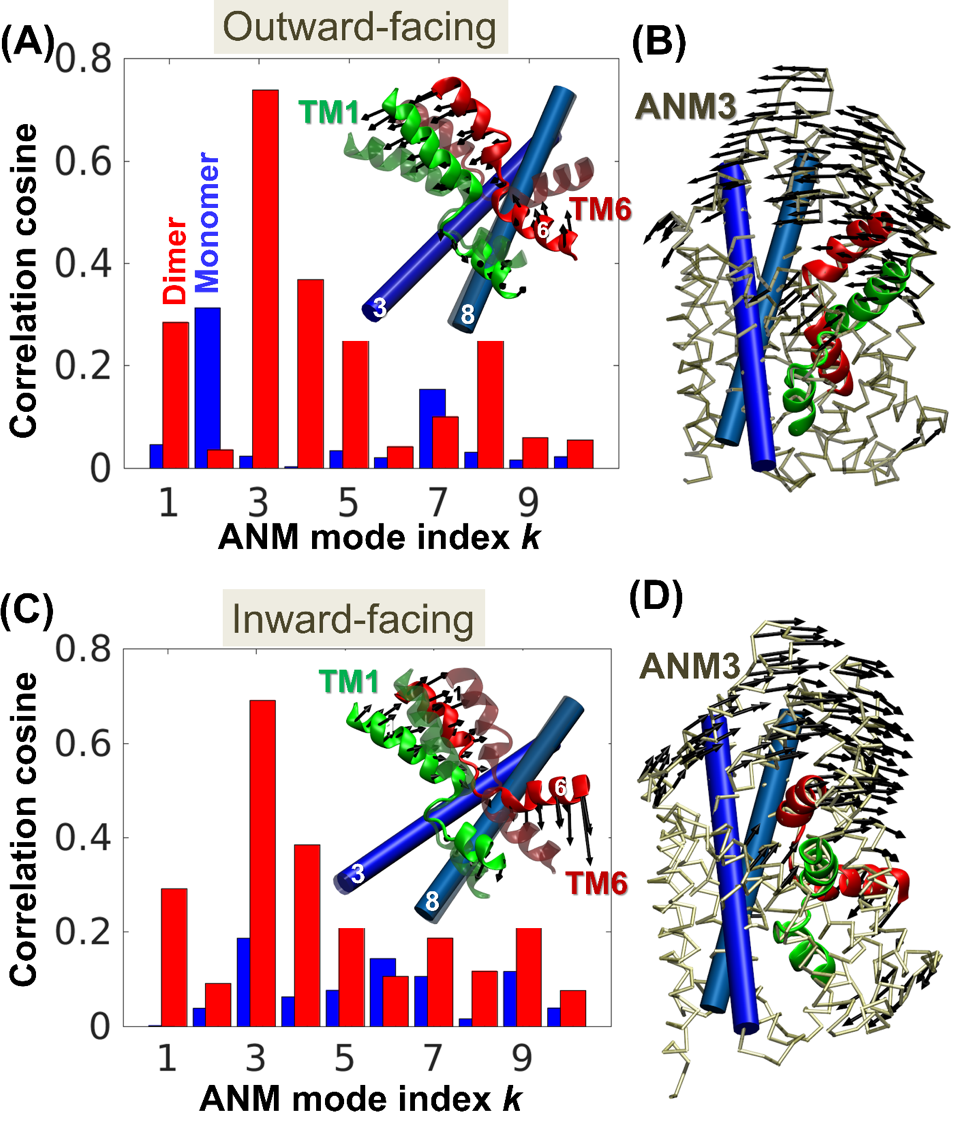
2.
Gur, M., Cheng, M. H., Zomot, E., Bahar, I. (2017).
Effect of dimerization on the dynamics of neurotransmitter: sodium symporters.
The Journal of Physical Chemistry B, 121(15), 3657-3666. (Joint corresponding author)
3.
Gur, M., Zomot, E., Cheng, M. H., Bahar, I. (2015).
Energy landscape of LeuT from molecular simulations.
The Journal of Chemical Physics, 143 (24), 243134.
4. Zomot, E.,
Gur, M., Bahar, I. (2015).
Microseconds simulations reveal a new sodium-binding site and the mechanism of sodium-coupled substrate uptake by LeuT.
Journal of Biological Chemistry, 290(1), 544-555.
5. Das, A.,
Gur, M., Cheng, M. H., Jo, S., Bahar, I., Roux, B. (2014).
Exploring the conformational transitions of biomolecular systems using a simple two-state anisotropic network model. PLOS Computational Biology, 10(4), e1003521.
Fundings:
Turkish Academy of Science (TÜBA)
Lab members involved:
Mert Gölcük ve Sema Zeynep Yılmaz